News Story
International Partnership Brings an Important Breakthrough in Fuel Cell Research
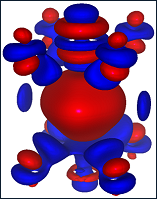
3D charge redistribution around the oxygen vacancy in BSCF perovskite with respect to neutral O atom. Blue and red isosurfaces (0.0025e/A3) correspond to excess and deficiency of the electron density, respectively. Oxygen vacancy (center) is surrounded by two B-metals - Co (top) and Fe (bottom), as well as four oxygen atoms from both sides.
Solid oxide fuel cells (SOFC) are promising devices generating energy in an environmentally friendly way, by a direct conversion of fossil and renewable fuels into electricity and high-quality heat. A major challenge in the SOFC improving is to reduce the operating temperature down to 500–600 C. This needs search of complex perovskites combining high O vacancy concentration (non-stoichiometry) and high vacancy mobility at moderate temperatures, as well as long-term cubic structure stability against transformation and degradation in polluted atmosphere. As we have demonstrated recently, the optimal strategy for developing new SOFC cathode should be based on the combination of key experimental data on oxygen incorporation into material and first-principles thermodynamics/kinetics of oxygen reduction and related processes. In this way, we were able to identify the atomistic mechanisms of key reactions for (La,Sr)MnO3 (LSM) with the emphasis on the rate-determining steps. Based on the present study, we plan to perform similar analysis for BSCF and related complex perovskites. This work is supported in part by NSF.
Published August 17, 2010









